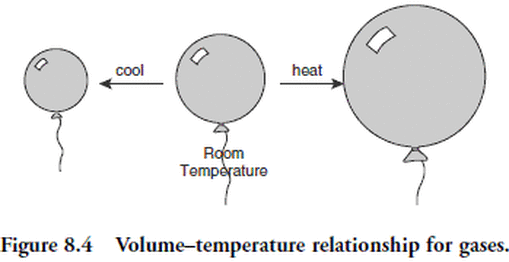
A third gas law may be derived as a corollary to Boyle's and Charles's laws. Suppose we double the thermodynamic temperature of a sample of gas. According to Charles’s law, the volume should double. Now, how much pressure would be required at the higher temperature to return the gas to its original volume? According to Boyle’s law, we would have to double the pressure to halve the volume. Thus, if the volume of gas is to remain the same, doubling the temperature will require doubling the pressure.This law was first stated by the Frenchman Joseph Gay-Lussac (1778 to 1850).
According to Gay-Lussac’s law:
"For a given amount of gas held at constant volume, the pressure is proportional to the absolute temperature."
Mathematically,
P1/T1=P2/T2 or P1*T2=P2*T1
P is pressure
T is temperature in kelvin
Gay-Lussac’s law tells us that it may be dangerous to heat a gas in a closed container. The increased pressure might cause the container to explode.
According to Gay-Lussac’s law:
"For a given amount of gas held at constant volume, the pressure is proportional to the absolute temperature."
Mathematically,
P1/T1=P2/T2 or P1*T2=P2*T1
P is pressure
T is temperature in kelvin
Gay-Lussac’s law tells us that it may be dangerous to heat a gas in a closed container. The increased pressure might cause the container to explode.