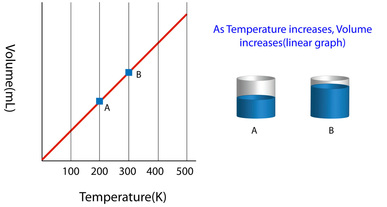
Charles's law (also known as the law of volumes) is an experimental gas law which describes how gases tend to expand when heated.
This law explains how a gas expands as the temperature increases; conversely, a decrease in temperature will lead to a decrease in volume. For comparing the same substance under two different sets of conditions, the law can be written as:
V1/T1=V2/T2
or
V1*T2=V2*T1
V is the volume of the gas
T is the temperature of the gas (measured in Kelvin).
k is a constant.
The equation shows that, as absolute temperature increases, the volume of the gas also increases in proportion. The law was named after scientist Jacques Charles, who formulated the original law in his unpublished work from the 1780s.
This law explains how a gas expands as the temperature increases; conversely, a decrease in temperature will lead to a decrease in volume. For comparing the same substance under two different sets of conditions, the law can be written as:
V1/T1=V2/T2
or
V1*T2=V2*T1
V is the volume of the gas
T is the temperature of the gas (measured in Kelvin).
k is a constant.
The equation shows that, as absolute temperature increases, the volume of the gas also increases in proportion. The law was named after scientist Jacques Charles, who formulated the original law in his unpublished work from the 1780s.

